 
ISO 13485 of the current 2016 version is based on an updated version of ISO 9001:2015, however, the structure of the standards’ sections differs from each other. ISO 13485 is an international industry standard developed by the International Organization for Standardization (ISO) in order to facilitate the implementation of harmonized established requirements into quality management systems and to ensure global harmonization of appropriate regulatory requirements for medical devices. Quality management systems for manufactures of medical devices Intellectual Property protection in Russia and foreign markets. 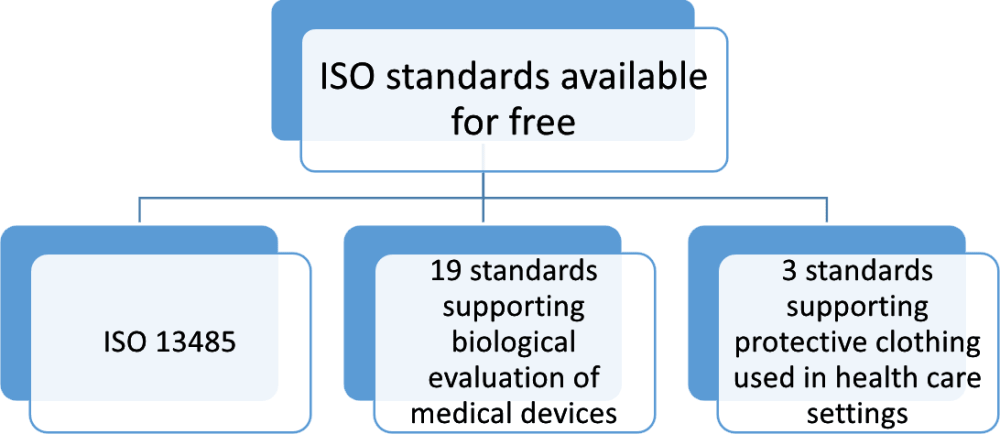
Aviation Engineering Certification Center.Evaluation of experience and business reputation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
